 This list is far from exhaustive and is in no particular order. This conclusion led to the theory that electrons exist in energy levels around the positive nucleus and have their own distinct properties in each of their energy levels. And so, without further ado, here are some must-know facts about Ernest Rutherford and his role in the foundation of nuclear physics. Laut Rutherford ist der Atomkern im Zentrum des Atoms sehr klein und positiv geladen. The technology did not exist then to investigate atoms in detail, so he imagined them as very tiny balls. This theory was adopted by Niels Bohr in 1913 who theorised that electrons could orbit the nucleus in a circular orbits and that the distance of the electron to the nucleus was fixed unless it moved between energy levels with the absorption or emission of light. In 1902 they formulated a revolutionary theory: that elements could disintegrate and be transformed into other elements. Das Rutherford Atommodell ist das erste Atommodell, das den Atomkern einführt. John Dalton (1766 1844) was an English scientist who developed an atomic theory early in the 19th century. Max Planck and Albert Einstein in the field of physics postulated that light energy can be absorbed and emitted as quanta. The small positive nucleus would deflect the few particles that came close. His model explained why most of the particles passed straight through the foil. During cathode ray tube experiment, a negatively charged particle was discovered by J.J. It was strongly supported by Sir Joseph Thomson, who had discovered the electron earlier. This model explained the description of an inner structure of the atom theoretically. The first part of his theory states that all matter is made of atoms, which are indivisible. Dalton based his theory on the law of conservation of mass and the law of constant composition. The electrons revolve in circular orbits about a massive positive charge at the centre. Thomson atomic model was proposed by William Thomson in the year 1900. Dalton's atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties. It was not until the earlier 20th Century that the scientific community arrived at the modern day atomic model. Rutherford proposed that the atom is mostly empty space. Now the atomic model had a central particle and electrons around it, reversing he plum pudding model of Thomson. An atom is the smallest unit of an element that can participate in a chemical change. Matter is composed of exceedingly small particles called atoms. Here are the postulates of Dalton’s atomic theory. He named this new fundamental particle as a proton. First published in 1807, many of Dalton’s hypotheses about the microscopic features of matter are still valid in modern atomic theory. 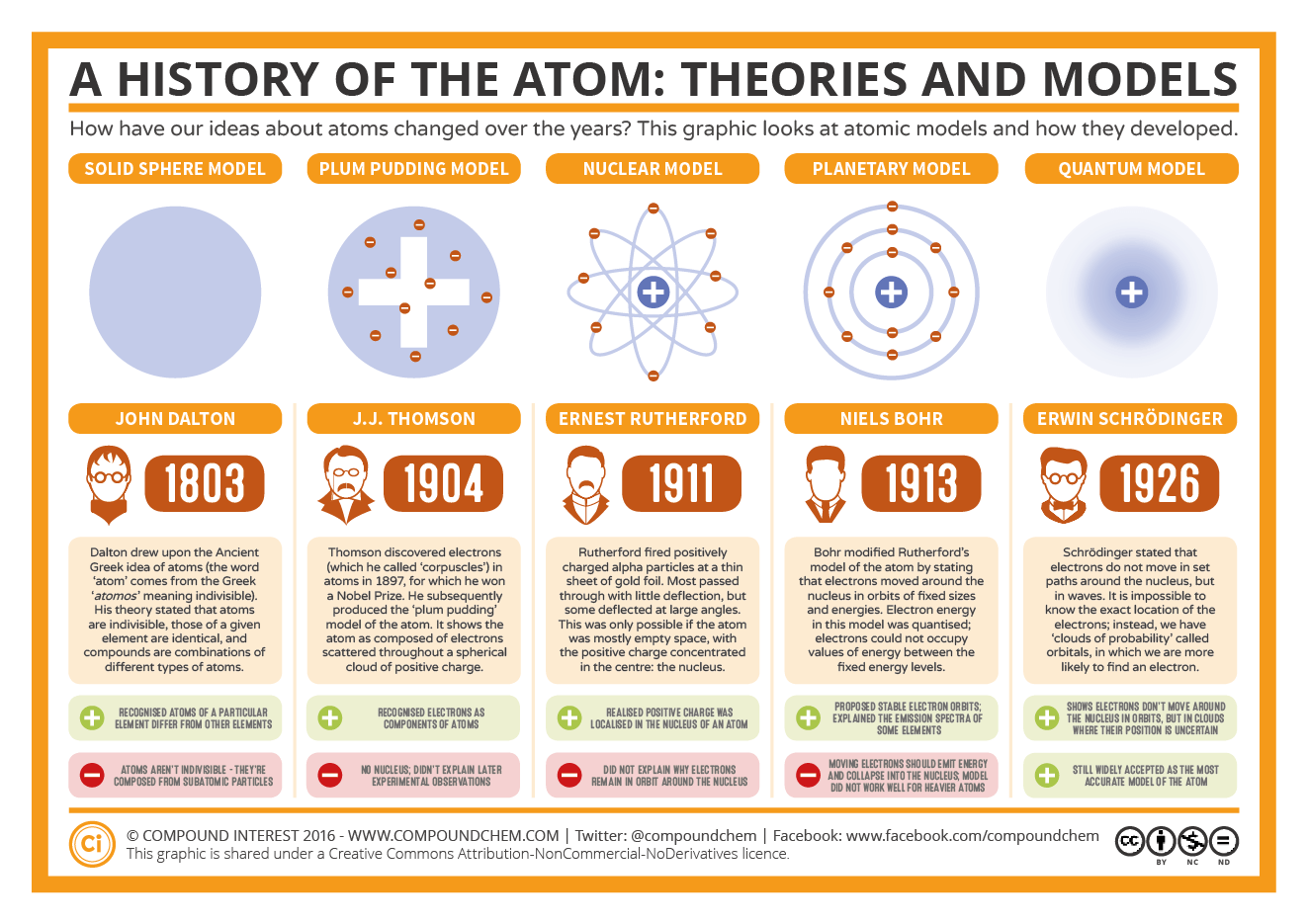
Rutherford conducted a number of experiments with hydrogen nuclei and nitrogen in air using alpha particles and after a number of theories concluded that the hydrogen atom made up other atoms. Rutherford further followed this up in 1917 when he proved that a hydrogen nucleus (1 proton) is present in other nuclei of different elements most notably nitrogen gas in the air. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
